ABSTRACT
Objective
To describe outcomes of patients with coronavirus disease 2019 (COVID-19) in the outpatient setting after early treatment with zinc, low dose hydroxychloroquine, and azithromycin (triple therapy) dependent on risk stratification.
Design
Retrospective case series study.
Setting
General practice.
Participants
141 COVID-19 patients with laboratory confirmed severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infections in the year 2020.
Main outcome measures
Risk-stratified treatment decision, rate of hospitalization and all-cause death.
Results
After 4 days (median, IQR 3-6, available for N=66/141) of onset of symptoms, 141 patients (median age 58 years, IQR 40-67; 73% male) received a prescription for the triple therapy for 5 days. Independent public reference data from 377 confirmed COVID-19 patients of the same community were used as untreated control. 4 of 141 treated patients (2.8%) were hospitalized, which was significantly less (p<0.001) compared with 58 of 377 untreated patients (15.4%) (odds ratio 0.16, 95% CI 0.06-0.5). One patient (0.7%) died in the treatment group versus 13 patients (3.5%) in the untreated group (odds ratio 0.2, 95% CI 0.03-1.5; p=0.12). There were no cardiac side effects.
Conclusions
Risk stratification-based treatment of COVID-19 outpatients as early as possible after symptom onset with the used triple therapy, including the combination of zinc with low dose hydroxychloroquine, was associated with significantly fewer hospitalizations.
1. INTRODUCTION
In December 2019, the new severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) started as an outbreak in Wuhan, China. This coronavirus has spread rapidly as a pandemic around the world [1], causing coronavirus disease 19 (COVID-19) pneumonia, acute respiratory distress syndrome (ARDS), cardiac injury, liver and renal injury, thrombosis, and death [2].
As of June 2020, diagnosis and treatment of COVID-19 have been almost exclusively studied from an inpatient perspective, including intensive care with mechanical ventilation. Only one study has described characteristics and key health outcomes of COVID-19 diagnosed patients in an outpatient setting [3]. This is surprising as primary care physicians often see COVID-19 patients first. They could play a critical role in early diagnosis, treatment, and management of disease progression and virus spread. This assumption is supported by the established principle in medicine that speed of eradication is linked to the outcome of life-threatening infections [4].
The early clinical phase of COVID-19 has not been the focus of much research so far, even though timing of antiviral treatment seems to be critical [5]. The optimal window for therapeutic intervention would seem to be before the infection spreads from upper to lower respiratory tract and before severe inflammatory reaction ensues [6]. Therefore, diagnosis and treatment of COVID-19 outpatients as early as possible, even based on clinical diagnosis only, may have been an underestimated first step to slow down or even stop the pandemic more effectively. Based on clinical application principles of antiviral therapies, as demonstrated in the case of influenza A [7], antiviral treatments should be used early in the course of infection.
Due to the lack of vaccines as well as SARS-CoV-2 specific therapies, the proposed use of repurposed antiviral drugs remains a valid practical consideration [8]. One of the most controversial drugs during the current SARS-CoV-2 pandemic is the well-known oral antimalarial drug hydroxychloroquine (HCQ), routinely used in the treatment of autoimmune diseases like rheumatoid arthritis or lupus [9, 10]. HCQ is currently listed as an essential medication for lupus by the World Health Organization (WHO) [11]. With more than 5.6 million prescriptions in the United States, HCQ was the 128th most commonly prescribed medication in 2017 [12]. In the meantime, first observational studies concluding beneficial therapeutic effects of HCQ as monotherapy or in combination with the antibiotic azithromycin were reported just a few weeks after the start of the SARS-CoV-2 outbreak [13]. All studies that used HCQ with rather contradictory results were done with hospitalized and often sicker patients [13], [14], [15], [16] and one publication was recently withdrawn [17, 18]. As of June 2020, no studies with COVID-19 outpatients treated with HCQ at an early stage of the disease have been reported.
Antiviral effects of HCQ are well-documented [19]. It is also known that chloroquine and probably HCQ have zinc ionophore characteristics, increasing intracellular zinc concentrations [20]. Zinc itself is able to inhibit coronavirus RNA-dependent RNA polymerase activity (RdRp) [21]. It has been hypothesized that zinc may enhance the efficacy of HCQ in treating COVID-19 patients [22]. The first clinical trial results confirming this hypothesis were recently published as preprint [23]. Nevertheless, many studies with HCQ in monotherapy or in combination with the antibiotic azithromycin have been inconclu
sive so far [13], [14], [15], [16]. In all of these studies, HCQ was used later than 5 days after onset of symptoms when hospitalized patients most likely had already progressed to stage II or III of the disease [6]. Regardless of the established antiviral effects of zinc and that many COVID-19 patients are prone to zinc deficiencies, dependent on comorbidities and drug treatments [22], none of these studies were designed to include zinc supplementation as combination treatment.
This first retrospective case series study with COVID-19 outpatients was done to show whether a) a simple to perform outpatient risk stratification might allow for rapid treatment decision shortly after onset of symptoms, and b) whether the triple 5-day therapy with zinc, low dose HCQ, and azithromycin might result in less hospitalizations and less fatalities compared with relevant public reference data of untreated patients.
2. METHODS
2.1. SETTING
This retrospective case series study analysed data from COVID-19 outpatients with confirmed severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection treated in a community in New York State, USA between March 18, 2020 and May 14, 2020. Outcome of patients who were treated with a specific triple therapy was compared to public reference data of patients in the same community who were not treated with this therapy.
2.2. CONFIRMATION OF COVID-19 DIAGNOSIS
COVID-19 diagnosis was confirmed if patients were positively tested for SARS-CoV-2 by means of PCR of nasal or pharyngeal swab specimens (majority of tests by Roche, Basel; 99,1% sensitivity and 99,7% specificity; other tests used with lower frequency included: Diasorin: 500 copies/mL; ThermoFisher: 10 genomic copy equivalents/reaction; Seegene: 1,250 copies/mL; Hologic: TCID50/mL: 1 × 10-2) or retrospectively by IgG detection tests (DiaSorin: Sensitivity 97.6% (≥ 15 days after diagnosis), specificity 99.3%; Diazyme: Sensitivity 91.2%, specificity 97.3%). Only patients who did have a record of a positive test result were included in the analysis. The PCR assays were authorized by the Food and Drug Administration (FDA) without clinical sensitivity/specificity data due to the urgent nature of the pandemic. Only one positive test was necessary for the patient to be included in the retrospective analysis.
2.3. PATIENTS
Sequentially consecutive COVID-19 outpatients older than 18 years at diagnosis were included in the analysis as treatment group. All patients were white. Patients received a prescription for the triple therapy only if they met one of the following risk stratification requirements during a medical office-based or telehealth consultation:
Group A: age >60 years; with or without clinical symptoms;
Group B: age ≤60 years and shortness of breath (SOB);
Group C: age ≤60 years, clinically symptomatic and with at least one of the following comorbidities: hypertension, hyperlipidemia, diabetes, obesity (body mass index ≥ 30 kg/m2), cardiovascular disease, heart failure, history of stroke, history of deep vein thrombosis or pulmonary embolism, asthma, chronic obstructive pulmonary disease (COPD), other lung disease, kidney disease, liver disease, autoimmune disease, or history of cancer. Pregnant women, if any, were to be included in this group as well.
Laboratory confirmed COVID-19 patients of the same community who were not treated with the described triple therapy and related outcome data represented the untreated control group which comprised both low risk and high risk patients (public reference data).
2.4. PROCEDURE AND TREATMENT
Data of treated patients were collected from electronic health records in the year 2020. Demographics, as reported by the patient, and a current medical history of hypertension, hyperlipidemia, diabetes, obesity (body mass index ≥ 30 kg/m2), cardiovascular disease, heart failure, stroke, asthma, COPD, other lung disease, kidney disease, liver disease, autoimmune disease, history of cancer, thyroid disease psychiatric disorder, or pregnancy were collected.
The presence of the following clinical symptoms of treated patients were documented: cough/dry cough, fever, SOB, changes of or no smell or taste, sore throat, headache, runny nose/clear rhinorrea, sinus congestion, diarrhea/vomiting, cold symptoms, feeling sick, weakness, and low back pain. If reported, number of days since onset of symptoms was documented.
The following vital signs, if available, were collected and documented: heart rate (beats per minute), breaths per minute (BPM), systolic and diastolic blood pressure (mmHg), body temperature (°C), oxygen saturation measured by pulse oximetry (O2 %), body weight (kg), and/or body mass index (BMI).
Main co-medications were characterised based on primary care prescriptions active at the time of diagnosis, were documented as categorical variables and included: beta-blockers, angiotensin-converting enzyme inhibitors, angiotensin-2 antagonists, calcium channel blockers, hydrochlorothiazide, statins, bronchodilators, antidiabetics, and insulin.
Only diagnosed COVID-19 patients who met the defined risk stratification requirements of group A, B, or C got a prescription for the following triple therapy for 5 consecutive days in addition to standard supportive care: zinc sulfate (220 mg capsule once daily, containing 50 mg elemental zinc), HCQ (200 mg twice daily), and azithromycin (500 mg once daily). No loading dose was used. Patients who did not meet the risk stratification requirements received standard of care to treat common upper respiratory infection. Patients were not treated with HCQ if they had known contraindications, including QT prolongation, retinopathy, or glucose-6-phosphate dehydrogenase (G6PD) deficiency. As usual and following best practice patients were informed about possible drug related side effects. Reported events, if any, were documented as required.
The selection of the used zinc supplement and drugs, dosages and the combination thereof, were based on treatment guidelines, positive reports from other countries like South Korea, emerging first clinical evidence, and based on the discretion of the treating physicians.
2.5. OUTCOME
Two outcomes were studied: COVID-19 related hospital admission and all-cause death during time of follow up of at least 28 days in the treatment group and in the untreated control group (public reference). The outcome of COVID-19 patients in the untreated control group was reported by the responsible health department.
2.6. STATISTICAL ANALYSES
Only patients in the treatment group who met the defined risk stratification requirements and who received at least a prescription for HCQ, with or without zinc, for 5 days, were included in the retrospective analysis and were cat
egorized accordingly. If the patient’s electronic health record did not include information on a clinical characteristic, it was assumed that the characteristic was not present. In the group of the public reference data only confirmed COVID-19 patients who were not treated in the respective general practice with the triple therapy were included in the analysis. For this untreated control group only outcome data for hospitalization and all-cause death was available and used for the statistical comparison with the treatment group.
No sample-size calculations were performed. Descriptive statistics are presented as median and interquartile range (IQR) for continuous variables and frequencies for categorical variables. For comparison with results of other studies means and standard deviations were calculated as needed. Normality of distribution for continuous variables was assessed by the Shapiro-Wilk-Test. A 2-tailed Student’s t-test was used for parametric analysis, and a Wilcoxon Signed-Rank test was used for nonparametric data analysis. For calculation of correlation the point biserial correlation coefficient was applied if one variable was dichotomous. Associations between two categorical variables were calculated with the Chi-Square test. Odds ratio (OR) were calculated for comparison of the outcome of the treatment group with the untreated control group. The α: 0.05 was considered as a significance level. The data were analysed using Microsoft Excel for Microsoft 365 MSO (32-Bit), the Excel add-on Real Statistics, SigmaStat 4, and Sigma Plot 14.0.
2.7. STUDY APPROVAL
The study was approved by the Western Institutional Review Board and it was exempt under 45 CFR § 46.104(d)(4). Reference number: D4-Excemption-Zelenko (06-16-2020). The analysis was conducted with de-identified patient data, according to the USA Health Insurance Portability and Accountability Act (HIPAA), Safe Harbor. For that reason exact dates and locations are not mentioned in this study.
3. RESULTS
3.1. PATIENTS
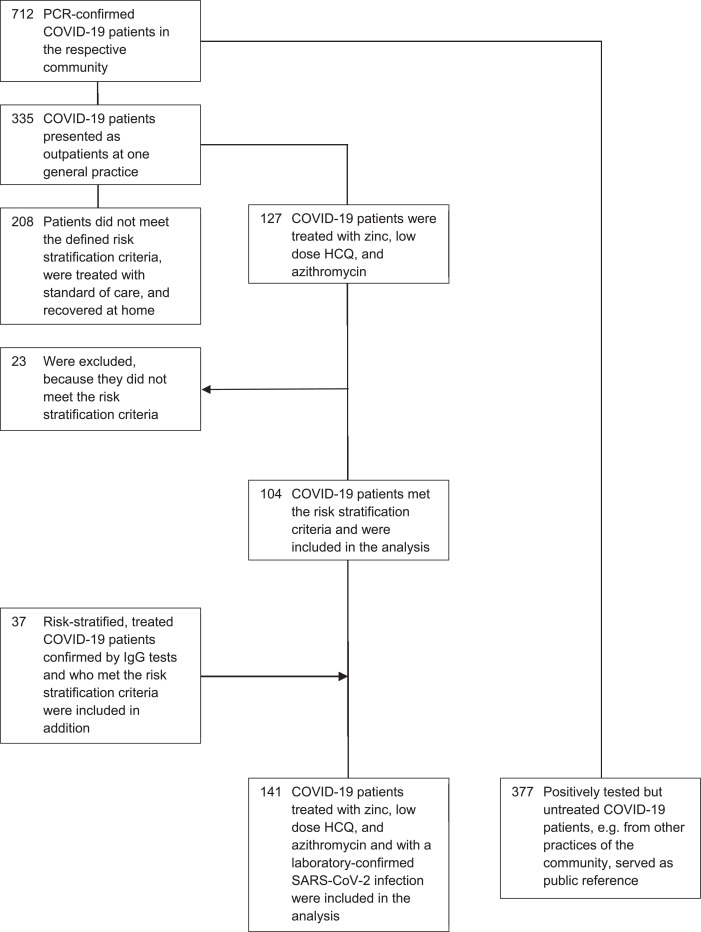
In accordance with available public reference data, 712 confirmed SARS-CoV-2 PCR positively tested COVID-19 patients were reported for the respective community at the defined time point of the analysis. Of these 712 patients, 335 presented as outpatients at a general practice and 127 were treated with the triple combination therapy. Of these 127 patients, 104 met the risk stratification criteria and were included in the analysis (table 1). 208 patients of the 335 did not meet the defined risk stratification criteria were treated with standard of care and recovered at home. The SARS-CoV-2 infection of 37 additional patients who were clinically diagnosed with COVID-19, who met the risk stratification criteria and who were also treated with the triple therapy, was later confirmed by IgG tests (table 1). These patients were included additionally in the analysis resulting in a total number of 141 patients, all with a confirmed SARS-CoV-2 infection by PCR or IgG tests. None of these patients were lost to follow-up for the defined outcome. The outcome of the remaining N=377 positively tested but not treated COVID-19 patients, e.g. from other practices of the community, served as public reference (fig 1). Analysis of the 141 patients in the treatment group showed that all of these patients (100%) got a prescription of HCQ, 136 (96.5%) of zinc sulfate, and 133 (94.3%) of azithromycin, while 1 patient (0.7%) got doxycycline instead. Instead of the triple therapy, 1 (0.7 %) patient in the treatment group got HCQ only, 7 (5%) patients got HCQ and zinc, and 4 (2.8%) patients got HCQ and azithromycin.
Table 1. COVID-19 Diagnostics by PCR and IgG tests of Patients in the Treatment Group
| COVID-19 Diagnostics no. (%) | Risk Stratified Group A (N=69) | Risk Stratified Group B (N=48) | Risk Stratified Group C (N=24) | All Patients Treatment Group (N=141) |
|---|---|---|---|---|
| SARS-CoV-2 – PCR Test | 51 (74) | 39 (81) | 14 (58) | 104 (74) |
| SARS-CoV-2 – IgG Test | 18 (26) | 9 (19) | 10 (42) | 37 (26) |
3.2. BASELINE CHARACTERISTICS OF THE PATIENTS
Table 2 shows the baseline demographics and clinical characteristics of all 141 patients in the treatment group and for the risk stratification groups A, B, and C. 69 patients (49%) belonged to group A, 48 (34%) to group B, and 24 (17%) to group C. Age ranged from 18 to 80 years and the median age was 58 years with an interquartile range (IQR) of 40-67. The median age of group A, B, and C was 67, 39, and 45 years. A total of 103 patients (73.1%) were male with a male-to-female ratio of 2.71. Most common comorbidities included hypertension (28%), obesity (28%), hyperlipidemia (23%), and diabetes (18%), whilst least common ones were liver disease (2%), heart failure (1%), and stroke (1%). One patient was pregnant (1%) at initiation of treatment. There was a positive and significant correlation between age and hypertension (r=0.3309, p=0.001), hyperlipidemia (r=0.26306, p<0.001), and cardiovascular disease (r=0.16757, p<0.05), while asthma was negatively correlated with age (r=-0.30867, p<0.001).
Table 2. Baseline Demographics and Clinical Characteristics of Patients in the Treatment Group*
| Characteristics | Risk Stratified Group A (N=69) | Risk Stratified Group B (N=48) | Risk Stratified Group C (N=24) | All Patients Treatment Group (N=141) |
|---|---|---|---|---|
| Median age (IQR) ̶ years | 67 (64-69) | 39 (24-47) | 45 (36-50) | 58 (40-67) |
| Male sex ̶ no. (%) | 46 (67) | 40 (83) | 17 (71) | 103 (73) |
| Coexisting conditions ̶ no. (%) | ||||
| Any condition | 44 (64) | 31 (65) | 24 (100) | 99 (70) |
| Hypertension | 27 (39) | 4 (8) | 8 (33) | 39 (28) |
| Hyperlipidemia | 21 (30) | 7 (15) | 5 (21) | 33 (23) |
| Diabetes | 16 (23) | 4 (8) | 5 (21) | 25 (18) |
| Obesity | 20 (29) | 10 (21) | 10 (42) | 40 (28) |
| Cardiovascular Disease | 9 (13) | 1 (2) | 3 (13) | 13 (9) |
| Heart Failure | 2 (3) | 0 (0) | 0 (0) | 2 (1) |
| Stroke | 1 (2) | 0 (0) | 0 (0) | 1 (1) |
| Asthma | 2 (3) | 9 (19) | 2 (8) | 13 (9) |
| COPD | 0 (0) | 0 (0) | 0 (0) | 0 (0) |
| Other Lung Disease | 6 (9) | 5 (10) | 4 (17) | 15 (11) |
| Kidney Disease | 1 (2) | 3 (6) | 2 (8) | 6 (4) |
| Liver Disease | 1 (2) | 2 (4) | 0 (0) | 3 (2) |
| Autoimmune Disease | 2 (3) | 4 (8) | 4 (17) | 10 (7) |
| History of Cancer | 6 (9) | 2 (4) | 1 (4) | 9 (6) |
| Thyroid Disease | 7 (10) | 4 (8) | 2(8) | 13 (9) |
| Psychiatric Disorder | 7 (10) | 4 (8) | 5 (21) | 16 (11) |
| Pregnancy | – | – | 1 (4) | 1 (1) |
- ⁎
-
IQR interquartile range
Median time between onset of clinical symptoms and medical consultation was 4 days (IQR 3-6; available for 66/141 patients, mean 4.8 days ± 2.7) (table 3). There was no significant correlation between age and days of onset of clinical symptoms to consultation (p>0.05). Days from onset of symptoms to consultation were not significantly different between groups (p>0.05).
Table 3. Patients with Reported Days Since Onset of Symptoms in the Treatment Group
| Characteristics | Risk Stratified Group A (N=69) | Risk Stratified Group B (N=48) | Risk Stratified Group C (N=24) | All Patients Treatment Group (N=141) |
|---|---|---|---|---|
| Patients with reported days ̶ no. (%) | 32 (46) | 25 (48) | 9 (38) | 66 (47) |
| Median days since onset of symptoms ̶ (IQR) | 4 (3-6) | 3 (3-6.5) | 4 (3-5.5) | 4 (3-6) |
Most common clinical symptoms included cough (87.2%), fever (77.3%), SOB (46.1%), and changes of or no smell or taste (30%), whilst least common ones were sinus congestion (16%), diarrhea/vomiting (5%), and low back pain (3%). Table 4 shows symptoms of all patients and stratified by groups A, B, and C. There was a significant negative correlation between age and changes of smell or taste (r=-0.43, p<0.001). No patient had a clinical diagnosis of pneumonia.
Table 4. COVID-19 Diagnostics and Baseline Reported Clinical Symptoms of Patients in the Treatment Group
| Clinical Symptoms ̶ no. (%) | Risk Stratified Group A (N=69) | Risk Stratified Group B (N=48) | Risk Stratified Group C (N=24) | All Patients Treatment Group (N=141) |
|---|---|---|---|---|
| Cough/Dry Cough | 60 (87) | 39 (81) | 24 (100) | 123 (87) |
| Fever | 53 (77) | 38 (79) | 18 (75) | 109 (77) |
| Shortness of Breath (SOB) | 17 (25) | 48 (100) | 0 (0) | 65 (46) |
| Changes of or no smell or taste | 21 (30) | 19 (40) | 2 (8) | 42 (30) |
| Sore Throat | 19 (28) | 8 (17) | 7 (29) | 34 (24) |
| Headache | 19 (28) | 6 (13) | 7 (29) | 32 (23) |
| Runny Nose/Clear Rhinorrhea | 16 (23) | 8 (17) | 4 (17) | 28 (20) |
| Sinus Congestion | 10 (15) | 9 (19) | 4 (17) | 23 (16) |
| Diarrhea/Vomiting | 1 (2) | 5 (10) | 1 (4) | 7 (5) |
| Cold Symptoms | 31 (45) | 16 (33) | 12 (50) | 59 (42) |
| Feels Sick | 40 (58) | 38 (79) | 17 (71) | 95 (67) |
| Weakness | 44 (64) | 22 (46) | 11 (46) | 77 (55) |
| Low Back Pain | 3 (4) | 0 (0) | 1 (4) | 4 (3) |
Table 5 shows vital signs, as they were available, for all patients and by group A, B, and C. Many patients consulted the general practice during the COVID-19 crisis via telehealth so vital signs were not available for all of these patients. The highest proportion of patients had available measurements for heart rate (63%) and pulse oximetry (60%). Vital signs were not significantly different between risk stratification groups (p>0.05) except for systolic blood pressure of group A and B (p<0.05).
Table 5. Physical Examination – Vital Signs of Patients in the Treatment Group
| Parameter | Patients with available Parameters ̶ no. (%) of N=141 | |
|---|---|---|
| Median Heart Rate ̶ beats per minute ̶ (IQR) | 86 (80-94) | 89 (63) |
| Median Breaths per Minute [BPM] ̶ (IQR) | 16 (15-18) | 43 (31) |
| Median Systolic Blood Pressure [mmHg] ̶ (IQR) | 126 (120-139) | 66 (47) |
| Median Diastolic Blood Pressure [mmHg] ̶ (IQR) | 80 (74-85.5) | 66 (47) |
| Median Body Temperature [°C] ̶ (IQR) | 37.2 (37-37.8) | 79 (56) |
| Median Pulse Oximetry [O2 %] ̶ (IQR) | 97 (96-98) | 85 (60) |
| Median Body Weight [kg] ̶ (IQR) | 88 (72.6-98.4) | 43 (31) |
| Median Body Mass Index [kg/m2] ̶ (IQR) | 32.2 (28.5-36.3) | 30 (21) |
Table 6 summarizes most important co-medications. 16% of patients were taking angiotensin-converting enzyme inhibitors, angiotensin-2-antagonists, hydrochlorothiazide or a combination thereof. The most common long-term therapies at the time of COVID-19 diagnosis were statins (20%), beta-blockers (12%), and insulin (18%). A few patients had chronic prescriptions for oral corticosteroids (9%), because of co-morbidities like asthma or autoimmune diseases and 3 patients (2%) got an additional antibiotic (levofloxacin) because of superinfections.
Table 6. Co-Medications of Patients in the Treatment Group
| Drug Class | Patients ̶ no. (%) of N=141 |
|---|---|
| Betablockers | 17 (12) |
| Angiotensin-converting enzyme inhibitors | 8 (6) |
| Angiotensin-2 Antagonists | 13 (9) |
| Calcium channel blockers | 8 (6) |
| Hydrochlorothiazide | 6 (4) |
| Statins | 28 (20) |
| Bronchodilators | 10 (7) |
| Antidiabetics | 11 (8) |
| Insulin | 26 (18) |
| Oral Corticosteroids | 13 (9) |
| Antibiotics | 3 (2) |
3.3. HOSPITALIZATIONS AND ALL-CAUSE DEATH
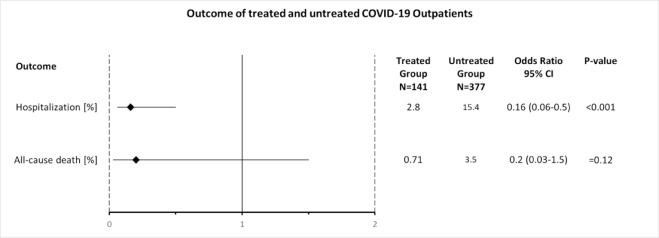
In the treatment group 4 of 141 patients were hospitalized, which was significantly less than in the untreated group with 58 of 377 patients (15.4%), (fig 2.), (OR 0.16; [95% CI, 0.06 to 0.5]; p<0.001), (table 7, fig 4). Therefore, the odds of hospitalization of treated patients were 84% less than in the untreated patients. All hospitalized patients were male, one in his twenties, two in their forties, and one in his seventies. Three of the 4 hospitalized patients (75%) belonged to risk stratification group B and one to group A (25%). All patients (100%) reported SOB at time of consultation. Median days from onset of symptoms to consultation were 4 days. Of the treatment group 1 patient had to stay only one day in hospital, 2 other patients were discharged as cured, and 1 patient died (s. below). No patient was on a ventilator.
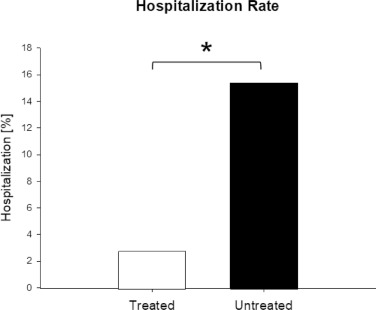
Table 7. Clinical Outcome in the Treated Patient Group versus the Untreated Patient Group
| Outcome | Treated Group ̶ no. (%) of N=141 | Untreated Group ̶ no. (%) of N=377 | Odds Ratio | 95% CI | P-value |
|---|---|---|---|---|---|
| Hospitalization | 4 (2.8) | 58 (15.4) | 0.16 | 0.06-0.5 | <0.001 |
| All-cause death | 1 (0.71) | 13 (3.5) | 0.2 | 0.03-1.5 | 0.12 |
CI=Confidence Interval
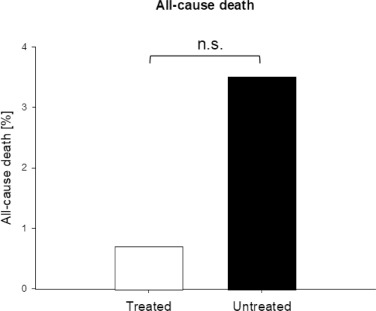
One of the 141 patients (0.71%) who belonged to treatment group A died after being hospitalized. This patient had a history of cancer and did only take one daily dose of the triple therapy before hospital admission. With 13 of 377 patients (3.5%, fig 3) more patients died in the untreated group (OR 0.2; [95% CI, 0.03 to 1.5]) (table 7, fig 4). Although the odds of all-cause death of treated patients were 80% less than in the untreated group, this difference did not reach statistical significance (p=0.12).
All patients of the treatment group with the clinical outcome hospitalization or all-cause death got a prescription for the complete triple therapy zinc, low dose hydroxychloroquine, and azithromycin.
The outcome of the 3 different risk-stratified groups A), B), and C) was not significantly different.
The 208 patients presenting at the general practice who did not meet the risk stratification requirements and who were not treated with the triple therapy recovered at home and no hospital admissions or deaths were reported.
3.4. SAFETY
In general, the triple therapy with zinc, low dose HCQ, and azithromycin was well tolerated. After initiation of treatment 30 of 141 patients (21%) reported weakness, 20 (14%) nausea, 15 (11%) diarrhea, and 2 (1%) rash (table 8). No patient reported palpitations or any cardiac side effect.
Table 8. Summary of Adverse Events
| Event | Patients ̶ no. (%) of N=141 |
|---|---|
| Any adverse event | 67 (48) |
| Weakness | 30 (21) |
| Nausea | 20 (14) |
| Diarrhea | 15 (11) |
| Rash | 2 (1) |
4. DISCUSSION
This first retrospective case series study with COVID-19 outpatients in primary care setting showed that risk-stratified treatment early after onset of clinical symptoms, with the triple therapy zinc, low dose HCQ, and azithromycin was associated with significantly less hospitalizations (odds ratio 0.16; p60 years and clinical symptoms), asthma and other lung diseases were the highest in group B (<60 years and SOB), and obesity and autoimmune disease were the highest in group C (<60 years, clinical symptoms, and defined comorbidities). Most frequent symptoms of these COVID-19 patients were cough followed by fever while available median body temperature measurements were in a normal range. Almost 50% of risk-stratified and treated patients were suffering from SOB while breaths per minute and blood oxygen saturation were still in the normal range. Median time from onset of symptoms to first medical consultation was 4 days (IQR 3-6). Approximately 16% of patients received co-medications known to be associated with zinc deficiency, such as antihypertensive drugs. No patient experienced any known severe adverse events that were considered drug related during treatment or follow up.
A growing number of reports provide evidence for for the effectiveness or otherwise of a range of COVID-19 drug treatments. Therefore, a living systematic review and network meta-analysis was published to assess how trustworthy the evidence is using the Grading of Recommendations Assessment, Development, and Evaluation (GRADE) approach [24]. Based on their most recent update from July 21, 2020 the authors conclude that glucocorticoids probably reduce mortality and mechanical ventilation in patients with COVID-19 compared with standard care. However, the effectiveness of most interventions is uncertain because most of the randomised controlled trials have been small so far and have important study limitations [24].
Another meta-analysis focused on the effectiveness of chloroquine derivatives in COVID-19 therapy [25]. The authors concluded that chloroquine derivatives are effective in improving clinical and virologic outcomes and may reduce mortality by a factor 3 in patients affected with COVID-19. They further conclude that big data are lacking basic treatment definitions and are subject of conflict of interest [25]. At the time of this manuscript submission, only one peer-reviewed study had analyzed the key health outcomes of COVID-19 patients diagnosed in primary care setting [3]. Because of this gap in data, the value of this study is multifold. It provides much needed recommendations for risk stratification and a treatment regimen to prevent hospitalization and death of COVID-19 patients. Diagnosis of COVID-19 for all patients in this analysis was confirmed by PCR or IgG tests compared with a recent study in which less than 3% had a diagnosis confirmed by laboratory tests [26]. To start the triple therapy as early as possible after symptom onset is critical for treatment success, because SARS-CoV-2 viral load seems to peak at day 5 to 6 after symptom onset [27], [28], [29] and severe cases progress to acute respiratory distress syndrome (ARDS) after only 8 to 9 days [30, 31]. Early antiviral treatment is an established protocol to manage severe disease progression, as was shown, for example, by a cumulative case control study during the 2009 H1N1 influenza pandemic in Canada [32]. For patients at high risk for severe viral disease progression, it is recommended to start antiviral therapy as early as possible [33, 34]. Early treatment might be also critically important to effectively reduce SARS-CoV-2 viral load [5], and this underscores the role of early intervention by primary care physicians as reported herein.
A further strength of this approach was the simple risk stratification of symptomatic outpatients to determine the need for therapy, a strategy not yet applied in COVID-19 primary care [35], but routinely implemented in primary care for other diseases [36]. Underlying assumptions of the risk stratification used in this setting are different than other recommendations [37]. Here, age stratified high risk was defined as >60 y
ears (typically defined as >65 years) to encompass the common increase of comorbidity incidences in this age group [38]. Patients ≤60 years with SOB, even without reduced pulse oximetry values, were treated because it was assumed virus will likely spread from upper to lower respiratory tract [39]. Also treated were patients ≤60 years with clinical symptoms and prognostically relevant comorbidities [37]. By applying this risk stratification approach, respective care was tailored to patients with a higher likelihood for hospitalizations or fatalities, which ensured that the medical principles of “patient first” and “doing no harm” were maintained [40]. As a result, 62% of COVID-19 patients were treated with standard of care only and recovered at home, and only 38% needed treatment with the triple therapy.
The antiviral potential of HCQ was broadly described in vitro and in vivo [41], [42], [43]. HCQ has a long terminal elimination half-life of 32 days in plasma and 50 days in blood [44]. Therefore, the treatment approach was conservative, with starting dose being the same as maintenance dose and with a short treatment duration of only 5 days, being even more conservative than other recommendations [42]. HCQ-dependent intracellular increases in pH might directly interfere with pH-dependent SARS-CoV-2 replication [19]. Also, chloroquine and probably HCQ have characteristics of a zinc ionophore resulting in increasing intracellular zinc concentrations [20]. The dose of elementary zinc in this study was similar to doses previously studied to successfully prevent infections in the elderly [45]. Antiviral effects of zinc against a variety of viruses have been demonstrated during the last decades [46]. Zinc, in addition to its role as a general stimulant of antiviral immunity, is known to specifically inhibit coronavirus RNA-dependent RNA polymerase [21]. Based on HCQ’s ionophore properties, it has been hypothesized that zinc may enhance the efficacy of HCQ in treating COVID-19 patients [22]. In addition, zinc might inhibit the serine protease furin [47]. Furin is expressed on endothelial cells, monocytes/macrophages, and smooth muscle cells in human atherosclerotic plaques [48] and therefore might play a critical role for the severe cardiovascular complications of COVID-19. As furin might be responsible to favor SARS-CoV-2 spreading compared with other beta coronaviruses [49, 50] and as furin-inhibition protects from certain viral-dependent infections [51], it may be important to evaluate the potential role of zinc in inhibiting this pathway.
Azithromycin was added to the treatment regimen as preliminary data provides evidence for more efficient or synergic virus elimination in conjunction with bacterial superinfection [13, 52]. Although there is a synergistic antiviral effect between zinc, HCQ, and azithromycin, zinc supplementation may be instrumental for the outcome of patient populations with severe clinical courses. Zinc deficiency was confirmed in a large number of healthy elderly [53] and in diabetic patients [54]. In addition, it has been documented that the antihypertensive drugs hydrochlorothiazide, angiotensin-converting-enzyme inhibitors, and angiotensin 2 receptor antagonists can result in an increased urinary excretion of zinc with subsequent systemic zinc deficiency [55]. Age, comorbidities, and relevant co-medications align well with the majority of described COVID-19 patients at high risk, including the risk-stratified population of this analysis. Zinc deficiency might explain why certain patient groups seem not to benefit from HCQ in monotherapy. During the 5-day treatment with the triple therapy and during follow up, no severe adverse events were observed and no cases of cardiac arrhythmia were reported in this general practice, which is in accordance with available safety data of more than 300,000 patients [56].
Inherent to all retrospective analyses, our study has certain limitations such as non-randomization and blinding of treatment. Also, only the outcome data of the untreated control group based on the public reference were available; because no other data on patient characteristics or clinical symptoms were available no risk adjustment was possible. Therefore, confounding factors and selection bias, among other issues, might exist. The demographic composition of the treatment group might have also had an influence on our findings. Because many physician appointments had to be managed by telehealth, vital parameters were not available for the majority of patients. Viral load and ECG data were not analyzed. Treatment with the triple therapy resulted in a numerically lower rate of all-cause deaths. In the absence of clinical details about the untreated patient group, the lower rate of all-cause death in the treated group was not statistically significant. However, the patients in the treated group were all positively risk-stratified while the risk of the untreated group was obviously lower as this group included high- and low-risk patients. When we compared the outcome of all risk-stratified patients in the study group (treated and non-treated) with the control patients (not stratified, treated with standard therapy), hospitalization and all-cause death were significantly less in the study group (p&
lt;0.0001 and p = 0.0154, respectively). These data were not shown in the results section because relevant clinical information was not completely available for all patients in the control group to allow risk adjustment between groups.
In this study, the ratio of males and average age was comparable with a relevant number of other studies, but distribution of comorbidities was not [57]. The latter was expected because outpatients usually have a different distribution of age and especially of comorbidities than critically ill inpatients. As expected the prevalence of hypertension, hyperlipidemia, and cardiovascular disease correlated positively with age while asthma correlated negatively. Approximately 50% of risk-stratified and treated patients presented with SOB while the parameters breaths per minute and blood oxygen saturation were still within the normal range. These patients would usually not be considered for hospital admission, although SOB might be considered an alarming early sign of disease progression. Based on the implemented risk stratification, these patients were identified and treated immediately.
Indeed, 3 of 4 hospitalized patients were in risk stratification group B including patients especially with SOB and also the hospitalized patient of group A reported SOB at time of consultation. This supports the assumption that COVID-19 patients with SOB are at much higher risk for disease progression and need to be monitored closely.
In contrast to many other studies, the most frequent symptom was cough and not fever [58, 59]. Changes in smell or taste in one third of patients and a negative correlation with age were similar to findings from other groups [60]. While mean time from onset of symptoms to treatment was only 4.8 days (median 4 days), previously reported time spans range from 6.3 days [61], to 8 days [16], up to 16.6 days [14], or it was often even not reported [62]. In most of these studies, COVID-19 disease had most likely already progressed at the time of presentation to stages II or even stage III of the disease [6]. In many studies, often only limited information is provided about co-medications and specifically about clinical symptoms at admission [62]. The latter would be very important to better understand the differences of clinical presentation between inpatients and outpatients, and thus the urgency for early anti-COVID-19 treatment in outpatient setting [63]. The potential of zinc to enhance the antiviral efficacy of HCQ was already described in detail elsewhere [22]. This hypothesis was recently confirmed by a study using a similar triple therapy and treatment duration [23]. Zinc added to HCQ and azithromycin resulted in a significantly increased number of patients being discharged, a reduction in mortality, or transfer to hospice. In another study, when a lower dose of 200 mg HCQ twice daily was added to basic treatment, mortality of even critically ill patients was significantly reduced [64]. These and our findings indicate that proper dosing of HCQ with its long half-life might be key for the favourable outcome of COVID-19 patients. In critical care, drugs with short half-lives are usually preferred. Especially in critically ill COVID-19 patients, higher doses of HCQ may have unforeseeable effects, for example, on insulin sensitivity in obese patients [65] and glucose levels in diabetics [66, 67]. Besides glucose levels, it is important to closely monitor renal function which is increasingly affected during progression of COVID-19 [68]. Because HCQ is substantially excreted by the kidneys, the risk of toxic reactions is greater in patients with impaired renal function [69].
4.1. POTENTIAL IMPLICATIONS FOR CLINICIANS AND POLICY MAKERS
Clinical experience from severely ill inpatients with pneumonia who were treated with high dose HCQ are not readily transferable to the outpatient setting with upper respiratory disease only. For outpatients with a median of only 4 days after onset of symptoms, COVID-19 represents a totally different disease and needs to be managed and treated differently [63]. A simple to perform outpatient risk stratification, as shown here, allows rapid treatment decisions and treatment with the triple therapy zinc, low dose HCQ, and azithromycin and may prevent a large number of hospitalizations and probably deaths during the SARS-CoV-2 pandemic. This might also help to avoid overwhelming of the health care systems.
Declaration of Competing Interest
The author Roland Derwand is/was at the time of writing an employee of Alexion Pharma Germany GmbH. His engagement and contribution to this study and publication was private and independent from his employer. The author Martin Scholz is/was at the time of writing External Senior Advisor for the company LEUKOCARE in Munich, Germany, and is/was Manging Director at Starts- and -Ups Consulting, Frankfurt, Germany. Vladmir Zelenko is/was general practitioner in New York State. All three authors confirm that this article content has no conflict of interest.
Acknowledgements
We thank all the patients and families involved in this study; the practitioners Dr. Rosy Joseph, Dr. Avery Knapp, Dr. Hillel Isseroff, Dr. William Grace, Dr. Sam Sandowski, and Dr. James Todaro for medical support; Chandra Duggirala, and Manoj Duggirala for operational and technical support; Mendel Mochkin (CrowdProtocol Foundation) for supporting the IRB submission; the reviewers Vjosa C. Mujko (Invivo Brands LLC) and Tzvi Jacobs who improved the language of this publication.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profi
t sectors.
Ethical Approval
The study was approved by the Western Institutional Review Board and it was exempt under 45 CFR § 46.104(d)(4). Ref. Number: D4-Excemption-Zelenko (06-16-2020)
All authors have completed the ICMJE uniform disclosure form at www.icmje.org/coi_disclosure.pdf and declare: no support from any organisation for the submitted work; no financial relationships with any organisations that might have an interest in the submitted work in the previous three years; no other relationships or activities that could appear to have influenced the submitted work.
REFERENCES
- [1]
- D. Wu, T. Wu, Q. Liu, et al.The SARS-CoV-2 outbreak: What we knowInt J Infect Dis, 94 (2020), pp. 44-48, 10.1016/j.ijid.2020.03.004[published Online First: 2020/03/17]ArticleDownload PDFView Record in ScopusGoogle Scholar
- [2]
- D. Atri, H.K. Siddiqi, J. Lang, et al.COVID-19 for the Cardiologist: A Current Review of the Virology, Clinical Epidemiology, Cardiac and Other Clinical Manifestations and Potential Therapeutic StrategiesJACC Basic Transl Sci, 5 (5) (2020), pp. 518-536, 10.1016/j.jacbts.2020.04.002[published Online First: 2020/04/16]ArticleDownload PDFView Record in ScopusGoogle Scholar
- [3]
- D. Prieto-Alhambra, E. Ballo, E. Coma-Redon, et al.Hospitalization and 30-day fatality in 121,263 COVID-19 outpatient casesmedRxiv (2020), 10.1101/2020.05.04.200900502020.05.04.20090050Google Scholar
- [4]
- D.A. Talan, J.J. Guterman, G.D. Overturf, et al.Analysis of emergency department management of suspected bacterial meningitisAnn Emerg Med, 18 (8) (1989), pp. 856-862, 10.1016/s0196-0644(89)80213-6[published Online First: 1989/08/01]ArticleDownload PDFView Record in ScopusGoogle Scholar
- [5]
- A. Gonçalves, J. Bertrand, R. Ke, et al.Timing of antiviral treatment initiation is critical to reduce SARS-Cov-2 viral loadmedRxiv (2020), 10.1101/2020.04.04.200478862020.04.04.20047886Google Scholar
- [6]
- H.K. Siddiqi, M.R. MehraCOVID-19 illness in native and immunosuppressed states: A clinical-therapeutic staging proposalJ Heart Lung Transplant, 39 (5) (2020), pp. 405-407, 10.1016/j.healun.2020.03.012[published Online First: 2020/05/05]ArticleDownload PDFV
iew Record in ScopusGoogle Scholar - [7]
- A.M. Fry, D. Goswami, K. Nahar, et al.Efficacy of oseltamivir treatment started within 5 days of symptom onset to reduce influenza illness duration and virus shedding in an urban setting in Bangladesh: a randomised placebo-controlled trialLancet Infect Dis, 14 (2) (2014), pp. 109-118, 10.1016/s1473-3099(13)70267-6[published Online First: 2013/11/26]ArticleDownload PDFView Record in ScopusGoogle Scholar
- [8]
- P. Schlagenhauf, M.P. Grobusch, J.D. Maier, et al.Repurposing antimalarials and other drugs for COVID-19Travel Med Infect Dis, 34 (2020), Article 101658, 10.1016/j.tmaid.2020.101658[published Online First: 2020/04/06]ArticleDownload PDFGoogle Scholar
- [9]
- D.J. WallaceThe use of chloroquine and hydroxychloroquine for non-infectious conditions other than rheumatoid arthritis or lupus: a critical reviewLupus, 5 (Suppl 1) (1996), pp. S59-S64[published Online First: 1996/06/01]View Record in ScopusGoogle Scholar
- [10]
- C. Gordon, M.B. Amissah-Arthur, M. Gayed, et al.The British Society for Rheumatology guideline for the management of systemic lupus erythematosus in adultsRheumatology (Oxford), 57 (1) (2018), pp. e1-e45, 10.1093/rheumatology/kex286[published Online First: 2017/10/14]CrossRefView Record in ScopusGoogle Scholar
- [11]
- (WHO) WHO. Model List of Essential Medicines 2020, https://list.essentialmeds.org/ [accessed 23 May 2020]Google Scholar
- [12]
- LLC C. Hydroxychloroquine Sulfate Drug Usage Statistics, United States, 2007 – 2017, 2020, https://clincalc.com/DrugStats/Drugs/HydroxychloroquineSulfate [accessed 23 May 2020]Google Scholar
- [13]
- Gautret P., Lagier J.C., Parola P., et al. Hydroxychloroquine and azithromycin as a treatment of COVID-19: results of an open-label non-randomized clinical trial. Int J Antimicrob Agents 2020:105949. doi: 10.1016/j.ijantimicag.2020.105949 [published Online First: 2020/03/25]Google Scholar
- [14]
- W. Tang, Z. Cao, M. Han, et al.Hydroxychloroquine in patients mainly with mild to moderate COVID-19: an open-label, randomized, controlled trialmedRxiv (2020), 10.1101/2020.04.10.200605582020.04.10.20060558Google Scholar
- [15]
- J. Chen, D. Liu, L. Liu, et al.A pilot study of hydroxychloroquine in treatment of patients with moderate COVID-19Zhejiang Da Xue Xue Bao Yi Xue Ban, 49 (2) (2020), pp. 215-219[published Online First: 2020/05/12]View Record in ScopusGoogle Scholar
- [16]
- M. Mahevas, V.-T. Tran, M. Roumier, et al.No evidence of clinical efficacy of hydroxychloroquine in patients hospitalized for COVID-19 infection with oxygen requirement: results of a study using routinely collected data to emulate a target trialmedRxiv (2020), 10.1101/2020.04.10.200606992020.04.10.20060699Google Scholar
- [17]
- Mehra M.R., Ruschitzka F., Patel A.N. Retraction—Hydroxychloroquine or chloroquine with or without a macrolide for treatment of COVID-19: a multinational registry analysis. The Lancet doi: 10.1016/S0140-6736(20)31324-6Google Scholar
- [18]
- The Lancet E. Expression of concern: Hydroxychloroquine or chloroquine with or without a macrolide for treatment of COVID-19: a multinational registry analysis. The Lancet doi: 10.1016/S0140-6736(20)31290-3Google Scholar
- [19]
- N. Yang, H.M. ShenTargeting the Endocytic Pathway and Autophagy Process as a Novel Therapeutic Strategy in COVID-19Int J Biol Sci, 16 (10) (2020), pp. 1724-1731, 10.7150/ijbs.45498[published Online First: 2020/04/01]CrossRefView Record in ScopusGoogle Scholar
- [20]
- J. Xue, A. Moyer, B. Peng, et al.Chloroquine is a zinc ionophorePLoS One, 9 (10) (2014), Article e109180, 10.1371/journal.pone.0109180[published Online First: 2014/10/02]CrossRefGoogle Scholar
- [21]
- A.J. te Velthuis, S.H. van den Worm, A.C. Sims, et al.Zn(2+) inhibits coronavirus and arterivirus RNA polymerase activity in vitro and zinc ionophores block the replication of these viruses in cell culturePLoS Pathog, 6 (11) (2010), Article e1001176, 10.1371/journal.ppat.1001176[published Online First: 2010/11/17]CrossRefGoogle Scholar
- [22]
- R. Derwand, M. ScholzDoes zinc supplementation enhance the clinical efficacy of chloroquine/hydroxychloroquine to win today’s battle against COVID-19?Med Hypotheses, 142 (2020), Article 109815, 10.1016/j.mehy.2020.109815[published Online First: 2020/05/15]ArticleDownload PDFGoogle Scholar
- [23]
- P. Carlucci, T. Ahuja, C.M. Petrilli, et al.Hydroxychloroquine and azithromycin plus zinc vs hydroxychloroquine and azithromycin alone: outcomes in hospitalized COVID-19 patientsmedRxiv (2020), 10.1101/2020.05.02.200800362020.05.02.20080036Google Scholar
- [24]
- R.A.C. Siemieniuk, J.J. Bartoszko, L. Ge, et al.Drug treatments for covid-19: living systematic review and network meta-analysisBMJ, 370 (2020), p. m2980CrossRefGoogle Scholar
- [25]
- Million M., Gautret P., Colson P., et al. Clinical Efficacy of Chloroquine derivatives in COVID-19 Infection: Comparative meta-analysis between the Big data and the real world. New Microbes and New Infections. https://doi.org/ 10.1016/j.nmni.2020.100709Google Scholar
- [26]
- D.R. Boulware, M.F. Pullen, A.S. Bangdiwala, et al.A Randomized Trial of Hydroxychloroquine as Postexposure Prophylaxis for Covid-19New England Journal of Medicine (2020), 10.1056/NEJMoa2016638Google Scholar
- [27]
- Y. Pan, D. Zhang, P. Yang, et al.Viral load of SARS-CoV-2 in clinical samplesLancet Infect Dis, 20 (4) (2020), pp. 411-412, 10.1016/s1473-3099(20)30113-4[published Online First: 2020/02/28]ArticleDownload PDFView Record in ScopusGoogle Scholar
- [28]
- J.Y. Kim, J.H. Ko, Y. Kim, et al.Viral Load Kinetics of SARS-CoV-2 Infection in First Two Patients in KoreaJ Korean Med Sci, 35 (7) (2020), p. e86, 10.3346/jkms.2020.35.e86[published Online First: 2020/02/23]CrossRefView Record in ScopusGoogle Scholar
- [29]
- L. Zou, F. Ruan, M. Huang, et al.SARS-CoV-2 Viral Load in Upper Respiratory Specimens of Infected PatientsN Engl J Med, 382 (12) (2020), pp. 1177-1179, 10.1056/NEJMc2001737[published Online First: 2020/02/20]CrossRefView Record in ScopusGoogle Scholar
- [30]
- C. Huang, Y. Wang, X. Li, et al.Clinical features of patients infected with 2019 novel coronavirus in Wuhan, ChinaLancet, 395 (10223) (2020), pp. 497-506, 10.1016/s0140-6736(20)30183-5[published Online First: 2020/01/28]ArticleDownload PDFView Record in ScopusGoogle Scholar
- [31]
- D. Wang, B. Hu, C. Hu, et al.Clinical Characteristics of 138 Hospitalized Patients With 2019 Novel Coronavirus-Infected Pneumonia in Wuhan, ChinaJama, 323 (11) (2020), pp. 1061-1069, 10.1001/jama.2020.1585[published Online First: 2020/02/08]CrossRefView Record in ScopusGoogle Scholar
- [32]
- R. Zarychanski, T.L. Stuart, A. Kumar, et al.Correlates of severe disease in patients with 2009 pandemic influenza (H1N1) virus infectionCmaj, 182 (3) (2010), pp. 257-264, 10.1503/cmaj.091884[published Online First: 2010/01/23]CrossRefView Record in ScopusGoogle Scholar
- [33]
- (CDC) CfDCaP. Influenza Antiviral Medications: Summary for Clinicians 2020, https://www.cdc.gov/flu/professionals/antivirals/summary-clinicians.htm [accessed 23 May 2020]Google Scholar
- [34]
- (CDC) CfDCaP. Managing People at High Risk for Severe Varicella 2020, https://www.cdc.gov/chickenpox/hcp/index.html?CDC_AA_refVal=https%3A%2F% [accessed 2020/05/23 2020]Google Scholar
- [35]
- Esper R.B., da Silva R.S., Oikawa F.T.C., et al. Empirical treatment with hydroxychloroquine and azithromycin for suspected cases of COVID-19 followed-up by telemedicine. In: Prevent Senior Institute SP, Brazil, ed. São Paulo, 2020:25Google Scholar
- [36]
- M. Johnson, L. Rigge, D. Culliford, et al.Primary care risk stratification in COPD using routinely collected data: a secondary data analysisNPJ Prim Care Respir Med, 29 (1) (2019), p. 42, 10.1038/s41533-019-0154-6[published Online First: 2019/12/05]View Record in ScopusGoogle Scholar
- [37]
- (CDC) CoDCaP. People Who Are at Higher Risk for Severe Illness 2020, https://www.cdc.gov/coronavirus/2019-ncov/need-extra-precautions/people-at-higher-risk.html [accessed 23 May 2020]Google Scholar
- [38]
- M. van den Akker, F. Buntinx, J.F. Metsemakers, et al.Multimorbidity in general practice: prevalence, incidence, and determinants of co-occurring chronic and recurrent diseasesJ Clin Epidemiol, 51 (5) (1998), pp. 367-375, 10.1016/s0895-4356(97)00306-5[published Online First: 1998/06/10]ArticleDownload PDFView Record in ScopusGoogle Scholar
- [39]
- G. Onder, G. Rezza, S. BrusaferroCase-Fatality Rate and Characteristics of Patients Dying in Relation to COVID-19 in ItalyJama (2020), 10.1001/jama.2020.4683[published Online First: 2020/03/24]Google Scholar
- [40]
- R.F. Ghaly, N.N. KnezevicWhat happened to “Patient first” and “Do no harm” medical principles?Surg Neurol Int, 9 (2018), p. 176, 10.4103/sni.sni_447_17[published Online First: 2018/09/18]CrossRefGoogle Scholar
- [41]
- J. Liu, R. Cao, M. Xu, et al.Hydroxychloroquine, a less toxic derivative of chloroquine, is effective in inhibiting SARS-CoV-2 infection in vitroCell Discov, 6 (2020), p. 16, 10.1038/s41421-020-0156-0[published Online First: 2020/03/21]CrossRefView Record in ScopusGoogle Scholar
- [42]
- X. Yao, F. Ye, M. Zhang, et al.In Vitro Antiviral Activity and Projection of Optimized Dosing Design of Hydroxychloroquine for the Treatment of Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2)Clin Infect Dis (2020), 10.1093/cid/ciaa237[published Online First: 2020/03/10]Google Scholar
- [43]
- S.A. Meo, D.C. Klonoff, J. AkramEfficacy of chloroquine and hydroxychloroquine in the treatment of COVID-19Eur Rev Med Pharmacol Sci, 24 (8) (2020), pp. 4539-4547, 10.26355/eurrev_202004_21038[published Online First: 2020/05/07]View Record in ScopusGoogle Scholar
- [44]
- S.E. Tett, D.J. Cutler, R.O. Day, et al.Bioavailability of hydroxychloroquine tablets in healthy volunteersBr J Clin Pharmacol, 27 (6) (1989), pp. 771-779, 10.1111/j.1365-2125.1989.tb03439.x[published Online First: 1989/06/01]CrossRefView Record in ScopusGoogle Scholar
- [45]
- A.S. Prasad, F.W. Beck, B. Bao, et al.Zinc supplementation decreases incidence of infections in the elderly: effect of zinc on generation of cytokines and oxidative stressAm J Clin Nutr, 85 (3) (2007), pp. 837-8
44, 10.1093/ajcn/85.3.837[published Online First: 2007/03/09]CrossRefView Record in ScopusGoogle Scholar - [46]
- S.A. Read, S. Obeid, C. Ahlenstiel, et al.The Role of Zinc in Antiviral ImmunityAdv Nutr, 10 (4) (2019), pp. 696-710, 10.1093/advances/nmz013[published Online First: 2019/07/16]CrossRefView Record in ScopusGoogle Scholar
- [47]
- P. Podsiadlo, T. Komiyama, R.S. Fuller, et al.Furin inhibition by compounds of copper and zincJ Biol Chem, 279 (35) (2004), pp. 36219-36227, 10.1074/jbc.M400338200[published Online First: 2004/05/14]View Record in ScopusGoogle Scholar
- [48]
- P. Stawowy, H. Kallisch, N. Borges Pereira Stawowy, et al.Immunohistochemical localization of subtilisin/kexin-like proprotein convertases in human atherosclerosisVirchows Arch, 446 (4) (2005), pp. 351-359, 10.1007/s00428-004-1198-7[published Online First: 2005/03/10]CrossRefView Record in ScopusGoogle Scholar
- [49]
- B. Coutard, C. Valle, X. de Lamballerie, et al.The spike glycoprotein of the new coronavirus 2019-nCoV contains a furin-like cleavage site absent in CoV of the same cladeAntiviral Res, 176 (2020), Article 104742, 10.1016/j.antiviral.2020.104742[published Online First: 2020/02/15]ArticleDownload PDFGoogle Scholar
- [50]
- J.K. Millet, G.R. WhittakerHost cell entry of Middle East respiratory syndrome coronavirus after two-step, furin-mediated activation of the spike proteinProc Natl Acad Sci U S A, 111 (42) (2014), pp. 15214-15219, 10.1073/pnas.1407087111[published Online First: 2014/10/08]CrossRefGoogle Scholar
- [51]
- S.A. Shiryaev, A.G. Remacle, B.I. Ratnikov, et al.Targeting host cell furin proprotein convertases as a therapeutic strategy against bacterial toxins and viral pathogensJ Biol Chem, 282 (29) (2007), pp. 20847-20853, 10.1074/jbc.M703847200[published Online First: 2007/06/01]View Record in ScopusGoogle Scholar
- [52]
- J. Andreani, M. Le Bideau, I. Duflot, et al.In vitro testing of combined hydroxychloroquine and azithromycin on SARS-CoV-2 shows syner
gistic effectMicrob Pathog, 145 (2020), Article 104228, 10.1016/j.micpath.2020.104228[published Online First: 2020/04/29]ArticleDownload PDFGoogle Scholar - [53]
- R.B. Ervin, J. Kennedy-StephensonMineral intakes of elderly adult supplement and non-supplement users in the third national health and nutrition examination surveyJ Nutr, 132 (11) (2002), pp. 3422-3427, 10.1093/jn/132.11.3422[published Online First: 2002/11/08]View Record in ScopusGoogle Scholar
- [54]
- R.A. Anderson, A.M. Roussel, N. Zouari, et al.Potential antioxidant effects of zinc and chromium supplementation in people with type 2 diabetes mellitusJ Am Coll Nutr, 20 (3) (2001), pp. 212-218, 10.1080/07315724.2001.10719034[published Online First: 2001/07/11]CrossRefView Record in ScopusGoogle Scholar
- [55]
- L.A. Braun, F. RosenfeldtPharmaco-nutrient interactions – a systematic review of zinc and antihypertensive therapyInt J Clin Pract, 67 (8) (2013), pp. 717-725, 10.1111/ijcp.12040[published Online First: 2013/01/03]CrossRefView Record in ScopusGoogle Scholar
- [56]
- J.C.E. Lane, J. Weaver, K. Kostka, et al.Safety of hydroxychloroquine, alone and in combination with azithromycin, in light of rapid wide-spread use for COVID-19: a multinational, network cohort and self-controlled case series studymedRxiv (2020), 10.1101/2020.04.08.200545512020.04.08.20054551Google Scholar
- [57]
- S. Richardson, J.S. Hirsch, M. Narasimhan, et al.Presenting Characteristics, Comorbidities, and Outcomes Among 5700 Patients Hospitalized With COVID-19 in the New York City AreaJama, 323 (20) (2020), pp. 2052-2059, 10.1001/jama.2020.6775[published Online First: 2020/04/23]CrossRefView Record in ScopusGoogle Scholar
- [58]
- A. Lovato, C. de FilippisClinical Presentation of COVID-19: A Systematic Review Focusing on Upper Airway SymptomsEar Nose Throat J (2020), 10.1177/0145561320920762145561320920762[published Online First: 2020/04/15]Google Scholar
- [59]
- T. Chen, D. Wu, H. Chen, et al.Clinical characteristics
of 113 deceased patients with coronavirus disease 2019: retrospective studyBmj, 368 (2020), p. m1091, 10.1136/bmj.m1091[published Online First: 2020/03/29]CrossRefGoogle Scholar - [60]
- A. Giacomelli, L. Pezzati, F. Conti, et al.Self-reported olfactory and taste disorders in SARS-CoV-2 patients: a cross-sectional studyClin Infect Dis (2020), 10.1093/cid/ciaa330[published Online First: 2020/03/28]Google Scholar
- [61]
- M. Million, J.C. Lagier, P. Gautret, et al.Early treatment of COVID-19 patients with hydroxychloroquine and azithromycin: A retrospective analysis of 1061 cases in Marseille, FranceTravel Med Infect Dis (2020), Article 101738, 10.1016/j.tmaid.2020.101738[published Online First: 2020/05/11]ArticleDownload PDFGoogle Scholar
- [62]
- J. Geleris, Y. Sun, J. Platt, et al.Observational Study of Hydroxychloroquine in Hospitalized Patients with Covid-19New England Journal of Medicine (2020), 10.1056/NEJMoa2012410Google Scholar
- [63]
- H.A. RischEarly Outpatient Treatment of Symptomatic, High-Risk Covid-19 Patients that Should be Ramped-Up Immediately as Key to the Pandemic CrisisAm J Epidemiol (2020), 10.1093/aje/kwaa093[published Online First: 2020/05/28]Google Scholar
- [64]
- Yu B., Li C., Chen P., et al. Low dose of hydroxychloroquine reduces fatality of critically ill patients with COVID-19. Sci China Life Sci 2020:1-7. doi: 10.1007/s11427-020-1732-2 [published Online First: 2020/05/18]Google Scholar
- [65]
- E. Mercer, L. Rekedal, R. Garg, et al.Hydroxychloroquine improves insulin sensitivity in obese non-diabetic individualsArthritis Res Ther, 14 (3) (2012), p. R135, 10.1186/ar3868[published Online First: 2012/06/09]CrossRefGoogle Scholar
- [66]
- A. Pareek, N. Chandurkar, N. Thomas, et al.Efficacy and safety of hydroxychloroquine in the treatment of type 2 diabetes mellitus: a double blind, randomized comparison with pioglitazoneCurr Med Res Opin, 30 (7) (2014), pp. 1257-1266, 10.1185/03007995.2014.909393[published Online First: 2014/03/29]CrossRefView Record in ScopusGoogle Scholar
- [67]
- A. GuptaReal-World Clinical Effectiveness and Tolerability of Hydroxychloroquine 400 Mg in Uncontrolled Type 2 Diabetes Subjects who are not Willing to Initiate Insulin Therapy (HYQ-Real-World Study)Curr Diabetes Rev, 15 (6) (2019), pp. 510-519, 10.2174/1573399815666190425182008[published Online First: 2019/11/13]CrossRefView Record in ScopusGoogle Scholar
- [68]
- C. Ronco, T. Reis, F. Husain-SyedManagement of acute kidney injury in patients with COVID-19Lancet Respir Med (2020), 10.1016/s2213-2600(20)30229-0[published Online First: 2020/05/18]Google Scholar
- [69]
- Plaquenil drug monographConcordia Pharmaceuticals Inc.U.S. Food & Drug Administration (FDA) (2017)https://www.accessdata.fda.gov/drugsatfda_docs/label/2017/009768s037s045s047lbl.pdf[accessed 4 June 2020]Google Scholar
- #
-
Derwand R and Scholz M contributed equally to the article.


